
Using RWD & Analytics to Measure the Economic Burden of Colorectal Cancer Complications
Colorectal cancer (CRC) is the third most commonly diagnosed cancer and the second leading cause of cancer deaths in both men and women.[1] According to estimates, there will be over 100,000 new cases of colon cancer, and over 40,000 cases of rectal cancer diagnosed in the US this year.[2]

ICER’s Development and Use of RWE: 5 Areas for Concern
The pervasiveness of real-world data (RWD) and the need to understand outcomes in clinical practice have galvanized the ubiquity and utility of real-world evidence (RWE). Especially within the life sciences industry, RWE is increasingly leveraged by research and development, epidemiology, outcomes research, and commercial groups across an asset’s[…]
ICER Takes Aim at Rising Drug Prices: 7 Things to Know
Written by Charles Tao. As an organization that evaluates the comparative value of medical technologies, the Institute for Clinical and Economic Review (ICER) is no stranger to ruffling the feathers of the life sciences industry. Its track record of declaring that drugs are overpriced says it all (see our recent blog post for a fuller[…]

Top 3 Questions Raised by ICER’s Proposed Changes to its Value Assessment Framework
Just 2 weeks ago, the Institute for Clinical and Economic Review (ICER) proposed changes to its 2017-2019 Value Assessment Framework, which guides the analytic structure of the primary domains of their framework. These 4 domains include comparative clinical effectiveness, long-term cost-effectiveness (CE), potential other benefits or disadvantages[…]

97% of Evaluated Drugs are Overpriced, According to ICER
2018 was not a good year for biopharma manufacturers, based on the Institute for Clinical and Economic Review’s (ICER) evaluation of interventions across various disease areas. Seemingly every month, ICER generated negative press for the biopharma industry. Headlines read:

BHE Holds Annual User Conference for its Instant Health Data (IHD) Platform
BHE hosted its annual Instant Health Data (IHD) User Conference on March 20, 2019 at the Four Seasons Hotel in Boston, Massachusetts. This day-long event brought together IHD users and analytics team leads from 20 top-ranked pharmaceutical companies, medical device manufacturers, and research organizations.
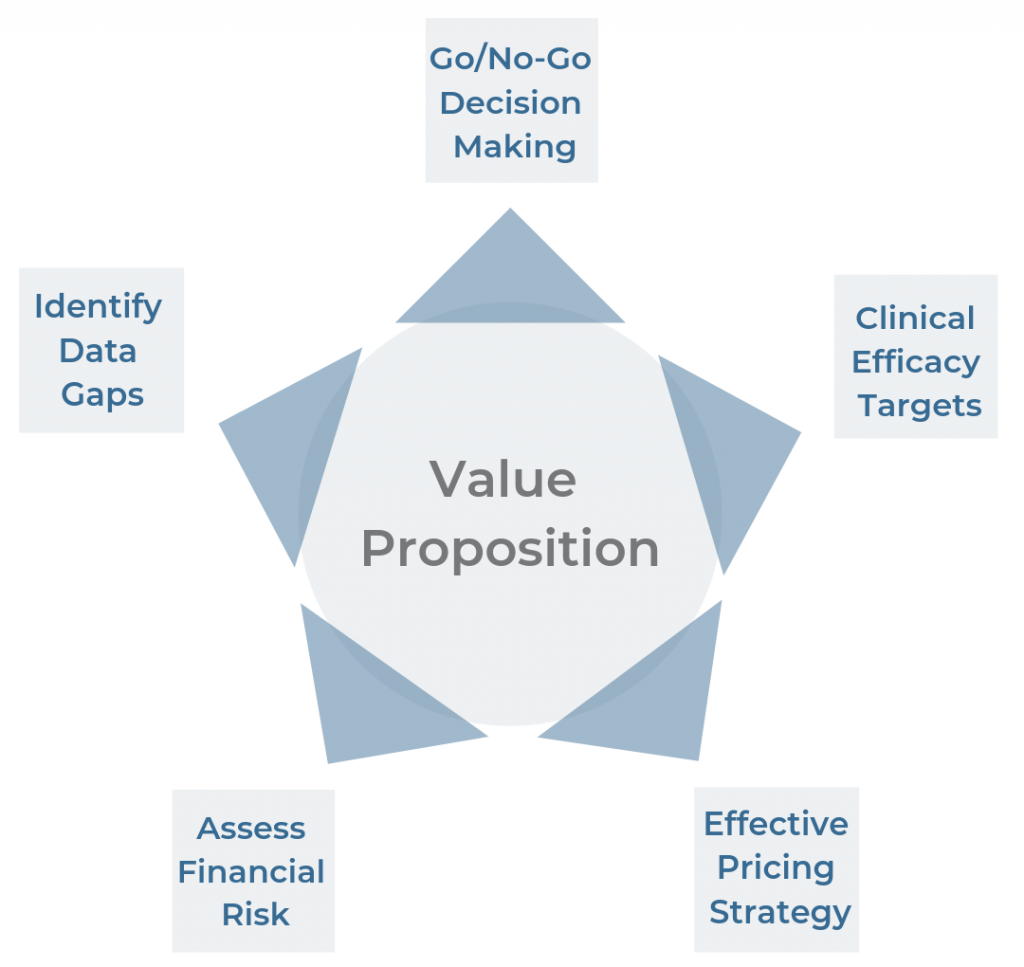
Five Ways to Use Early-Stage Models to Support Your Drug’s Value Proposition
Written by Jennifer Benner. In my previous post, I explored the differences between early- and late-stage economic modeling, including a discussion of the benefits of using early-stage models. In this post, I will examine 5 ways that utilizing early-stage models can benefit drug manufacturers and help produce more efficient, cost-effective drug[…]
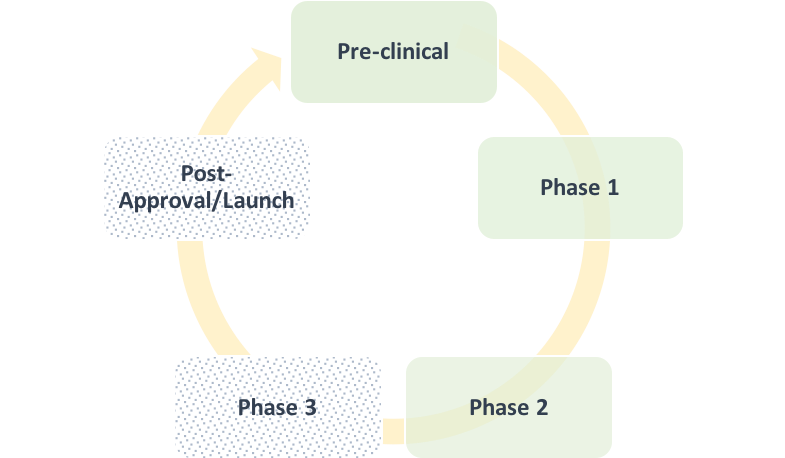
How to Predict if a Drug will be Cost-Effective: A Case for Early-Stage Modeling
Written by Jennifer Benner. Wouldn’t it be nice if there were a crystal ball that could predict, during the very early stages of drug development, whether a new medication would represent good value for money at its intended launch price? Or which efficacy targets it needed to hit in order to remain cost-effective? Or what an assessment by the[…]
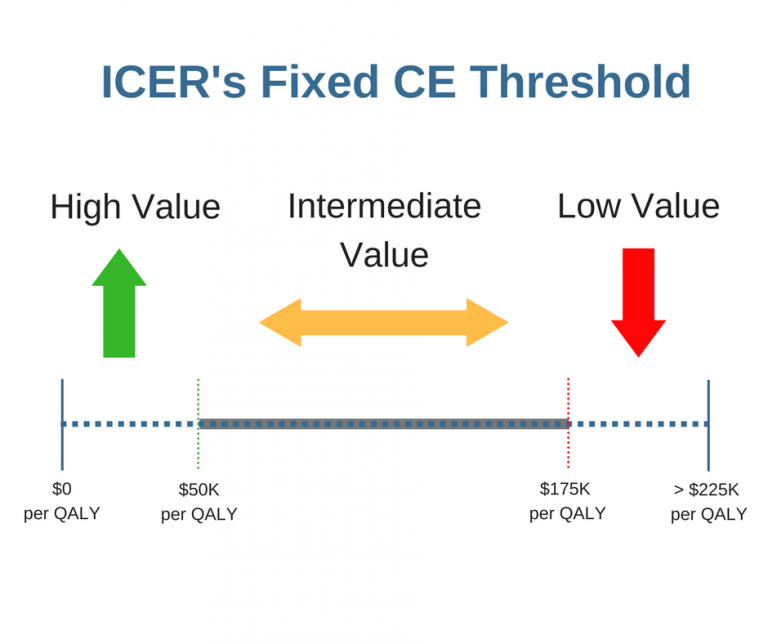
Do Research Groups Align on the Value of an Intervention? A Prelude to our ISPOR Barcelona Presentation
Do Research Groups Align on the Value of an Intervention? A Prelude to our ISPOR Barcelona Presentation By Matthew Sussman

Instant Health Data User Conference Showcases Customer-Driven Applications of Real-World Evidence
BHE held its 2018 Instant Health Data (IHD) User’s Conference on Wednesday, March 14th at the Ritz Carlton Hotel in Philadelphia, Pennsylvania. Real-world evidence (RWE) managers and IHD users attended from over a dozen life science companies and payers, representing about one-half of all companies licensing IHD. These researchers regularly use[…]
