-1.png)
The Use of RWE in ICER Evaluations: What It Means for Life Sciences Companies
Real-world evidence (RWE) has been gaining acceptance at a rapid rate in many areas of the life sciences industry including R&D, epidemiology, outcomes research, and commercial groups. The COVID-19 pandemic and resulting research to find treatments and vaccines has accelerated the growth and adoption of RWE as well. The Institute for Clinical and[…]

You Can Influence ICER Recommendations, You Just Need the Right Evidence
It’s difficult to dispute the findings of an advisory group which is widely accepted as the authority, especially when it comes to evaluating the value of different therapeutic interventions. The cost of drugs in the US has been a hot button topic in recent years, so healthcare industry stakeholders are increasingly looking for objective[…]

ICER’s Development and Use of RWE: 5 Areas for Concern
The pervasiveness of real-world data (RWD) and the need to understand outcomes in clinical practice have galvanized the ubiquity and utility of real-world evidence (RWE). Especially within the life sciences industry, RWE is increasingly leveraged by research and development, epidemiology, outcomes research, and commercial groups across an asset’s[…]
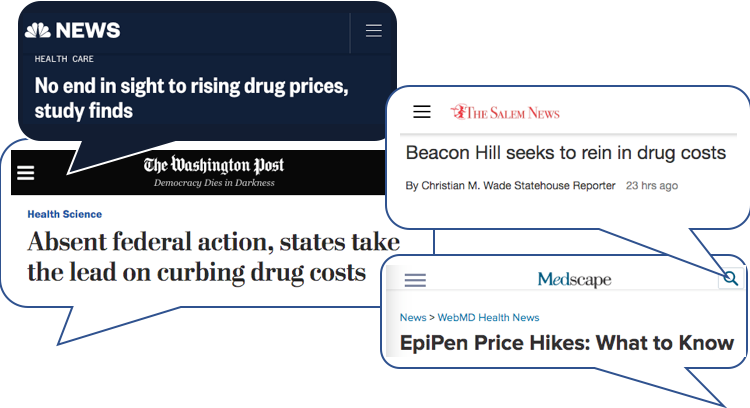
ICER Takes Aim at Rising Drug Prices: 7 Things to Know
Written by Charles Tao. As an organization that evaluates the comparative value of medical technologies, the Institute for Clinical and Economic Review (ICER) is no stranger to ruffling the feathers of the life sciences industry. Its track record of declaring that drugs are overpriced says it all (see our recent blog post for a fuller[…]

Top 3 Questions Raised by ICER’s Proposed Changes to its Value Assessment Framework
Just 2 weeks ago, the Institute for Clinical and Economic Review (ICER) proposed changes to its 2017-2019 Value Assessment Framework, which guides the analytic structure of the primary domains of their framework. These 4 domains include comparative clinical effectiveness, long-term cost-effectiveness (CE), potential other benefits or disadvantages[…]

97% of Evaluated Drugs are Overpriced, According to ICER
2018 was not a good year for biopharma manufacturers, based on the Institute for Clinical and Economic Review’s (ICER) evaluation of interventions across various disease areas. Seemingly every month, ICER generated negative press for the biopharma industry. Headlines read:
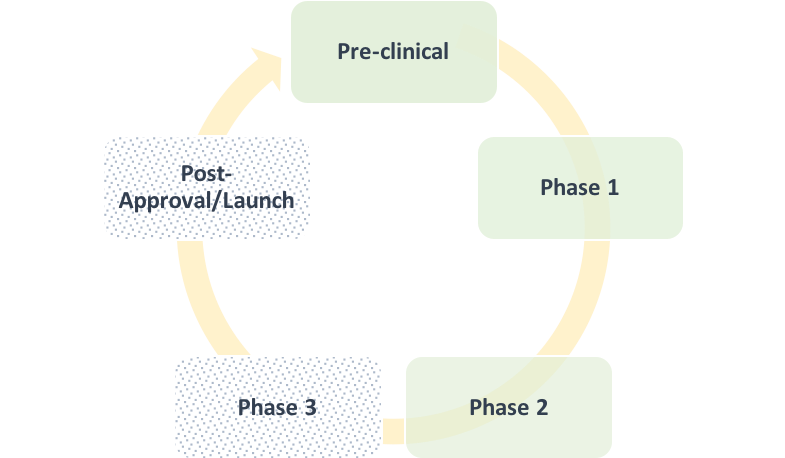
How to Predict if a Drug will be Cost-Effective: A Case for Early-Stage Modeling
Written by Jennifer Benner. Wouldn’t it be nice if there were a crystal ball that could predict, during the very early stages of drug development, whether a new medication would represent good value for money at its intended launch price? Or which efficacy targets it needed to hit in order to remain cost-effective? Or what an assessment by the[…]
Do Research Groups Align on the Value of an Intervention? A Prelude to our ISPOR Barcelona Presentation
Do Research Groups Align on the Value of an Intervention? A Prelude to our ISPOR Barcelona Presentation By Matthew Sussman
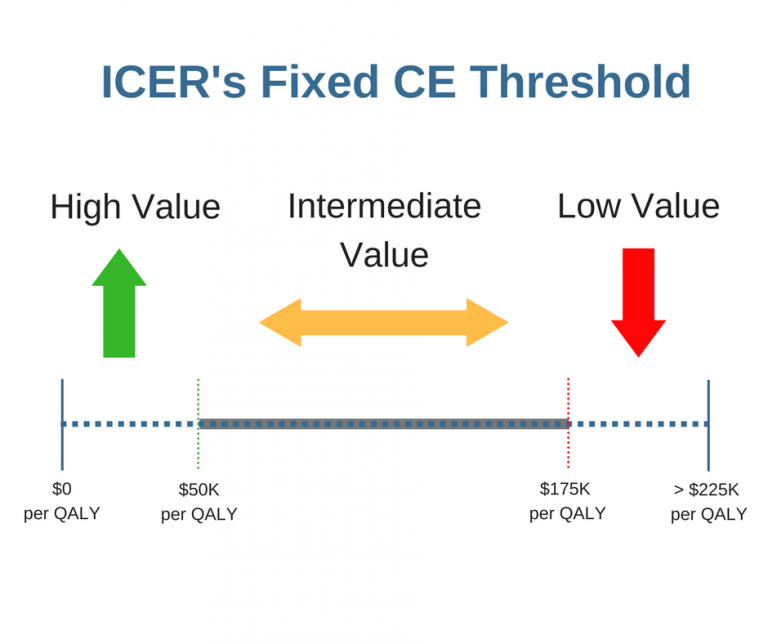
ICER Versus NICE: Who Cares About Value-Based Pricing Assessments?
This is my last post in an ICER versus NICE series that examines how ICER’s and NICE’s methodologies for implementing cost-effectiveness (CE) analyses differ. My last post explored how differences in the use of a fixed CE threshold and the application of a narrower CE threshold range may lead to different intervention recommendations. In this[…]
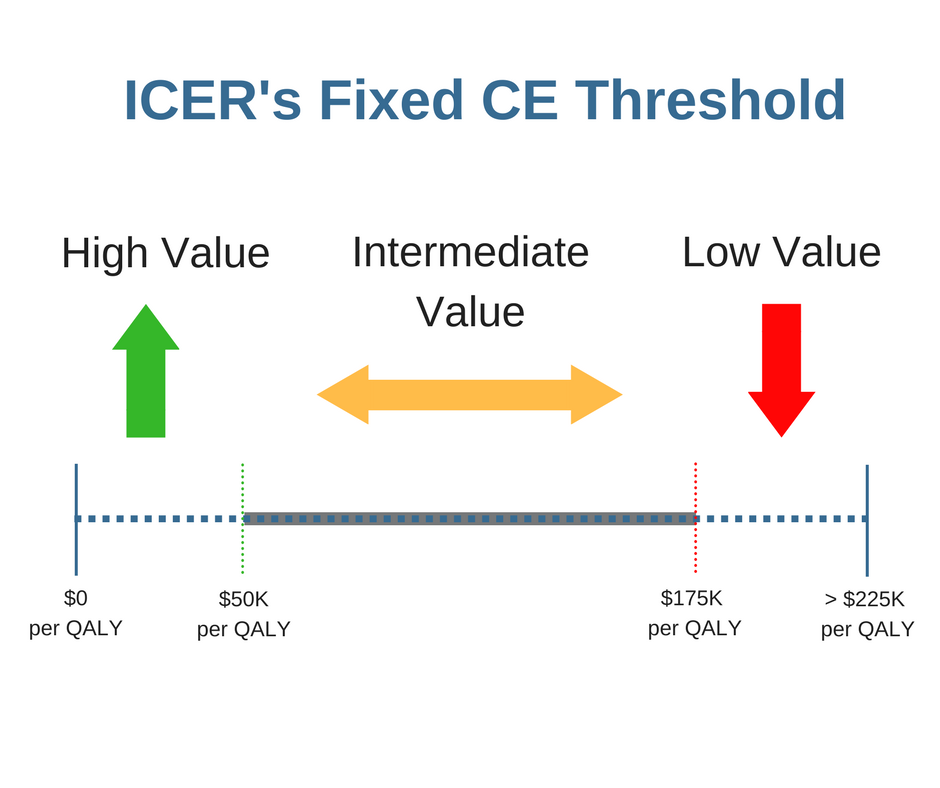
Do Fixed Cost-Effectiveness Thresholds or CE Threshold Magnitudes Matter?
In my last post, I highlighted three subtle differences between ICER’s and NICE’s methodologies when conducting cost-effectiveness (CE) analyses. These differences, if overlooked, could actually lead to very different recommendations for coverage, reimbursement, and pricing. Two of these important differences revolve around CE thresholds,[…]
